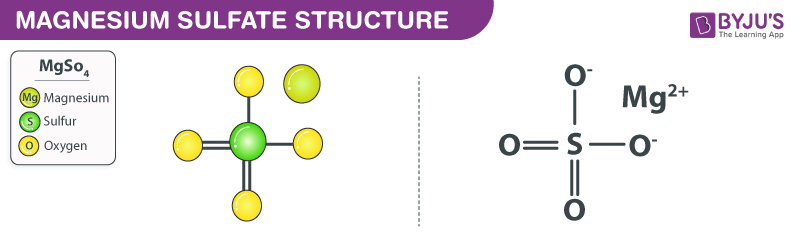
The Bonds in the Compound Mgso4 Can Be Described as
The number of orbitals in each subshell for the first four shells is summarised in Table 171. A d-subshell has five orbitals and an f-subshell has seven.

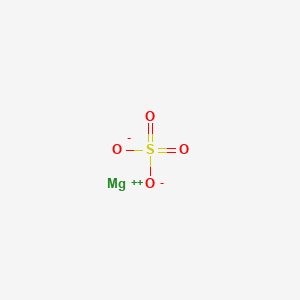
Is Mgso4 Magnesium Sulfate Ionic Or Covalent Youtube
An s-subshell has just one orbital.

. Orbitals can be described as regions of space surrounding the nucleus of an atom in which electrons may be found. The total number of orbitals in a shell is. A p-subshell has three orbitals.
P P3- IONIC COMPOUNDS metal with nonmetal ion - ion Caion anion Predicting Oxidation Number or Charge from Periodic Table Forming Cations metals lose e- form cations oxidation group 1A lose 1 1 charge 1 group 2A lose 2 2 charge 2 group 3A lose 3 3 charge 3 group 4A lose 4 4 charge 4 Forming Anions Non-metals gain e- form anions Acids Bases and.

How To Draw The Lewis Dot Structure For Mgso4 Magnesium Sulfate Youtube
Comments
Post a Comment